These elements are characterized by having three valence electrons.Boron is a metalloid, it has characteristics intermediate between metals and non-metals, and the rest of the group are metals. In this group, we begin to see the changeover toward the non-metallic character.Properties for Group 13 of Periodic Table elements Group 13 of the Periodic Table consists of the elements boron (B), aluminum (Al), gallium (Ga), indium (In), and thallium (Tl). They react readily with halogens to form ionic salts and can react slowly with water.Beryllium is the least metallic element in the group and tends to form covalent bonds in its compounds.These elements all have two valence electrons and tend to lose both to form ions with a two-plus charge.The alkaline earth metals are silvery-coloured, soft, low-density metals, though are a bit harder than the alkali metals.Properties for Group 2 of Periodic Table Elements Group 2 of Periodic Table The series consists of the elements beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). They react with water to liberate hydrogen gas.Īlkali metal + water → Alkali metal hydroxide + hydrogen.The alkali metals react readily with halogens to form ionic salts, such as table salt, and sodium chloride (NaCl).Due to their activity, they occur naturally in ionic compounds, not in their elemental state.This makes them very reactive and they are the most active metals. They have the lowest ionization energies in their respective periods. 
These elements all have one valence electron which is easily lost to form an ion with a single positive charge.The alkali metals are silver-colored (cesium has a golden tinge), soft, low-density metals.However, even these small quantities are required for the body to function properly. These last three metals are present in the body in very small quantities. Cobalt is a necessary component of vitamin B-12, a vital nutrient. Manganese is needed for the body to metabolize oxygen properly. Copper is also needed for several proteins to function properly in the body. 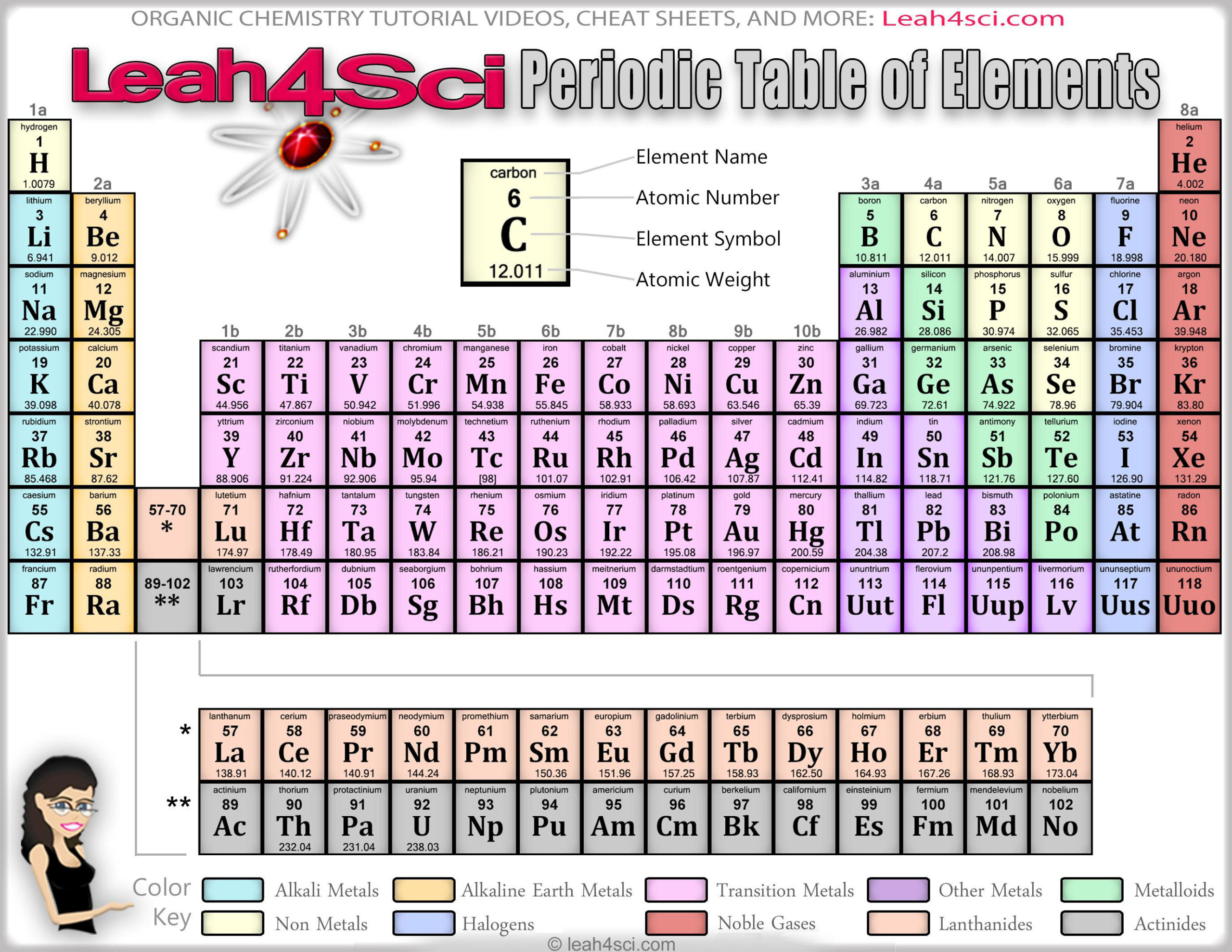
Zinc is needed for the body’s immune system to function properly, as well as for protein synthesis and tissue and cell growth. Other transition metals have important functions in the body, despite being present in low amounts. It is the presence of this particular transition metal in your red blood cells that allows you to use the oxygen you inhale. Each hemoglobin molecule has four iron atoms, which act as binding sites for oxygen. The crucial atom in the hemoglobin protein is iron. Hemoglobin is a relatively large molecule, with a mass of about 65,000 u. Hemoglobin combines with oxygen and carbon dioxide, transporting these gases from one location to another in the body. The critical part of the red blood cell is a protein called hemoglobin. Without red blood cells, animal respiration as we know it would not exist. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs. Iron is a transition metal and the chemistry of iron makes it a key component in the proper functioning of red blood cells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The most abundant non-main group element is iron, at 0.006 percentage by mass. Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. 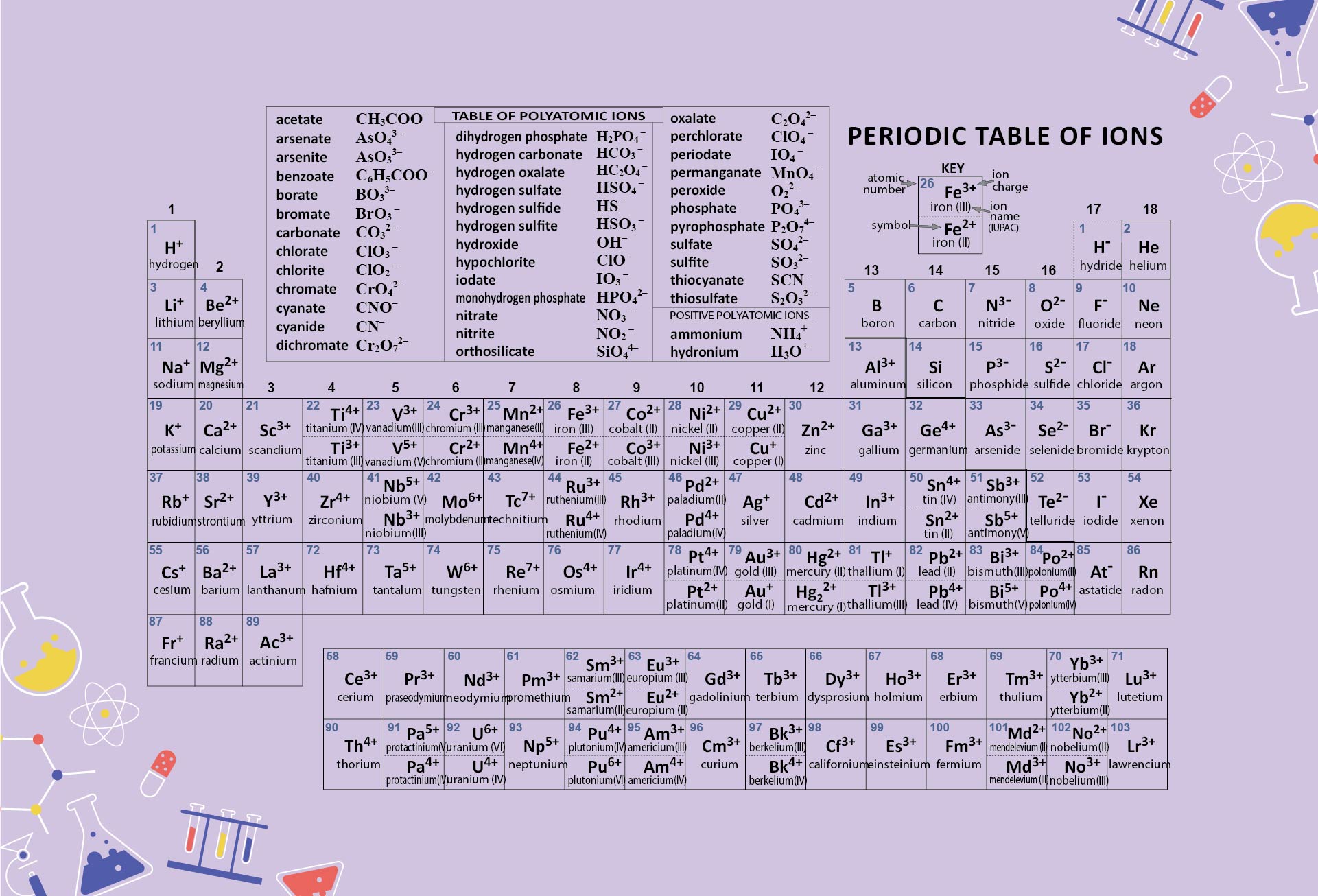
Elements are either metals, nonmetals, or semimetals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
